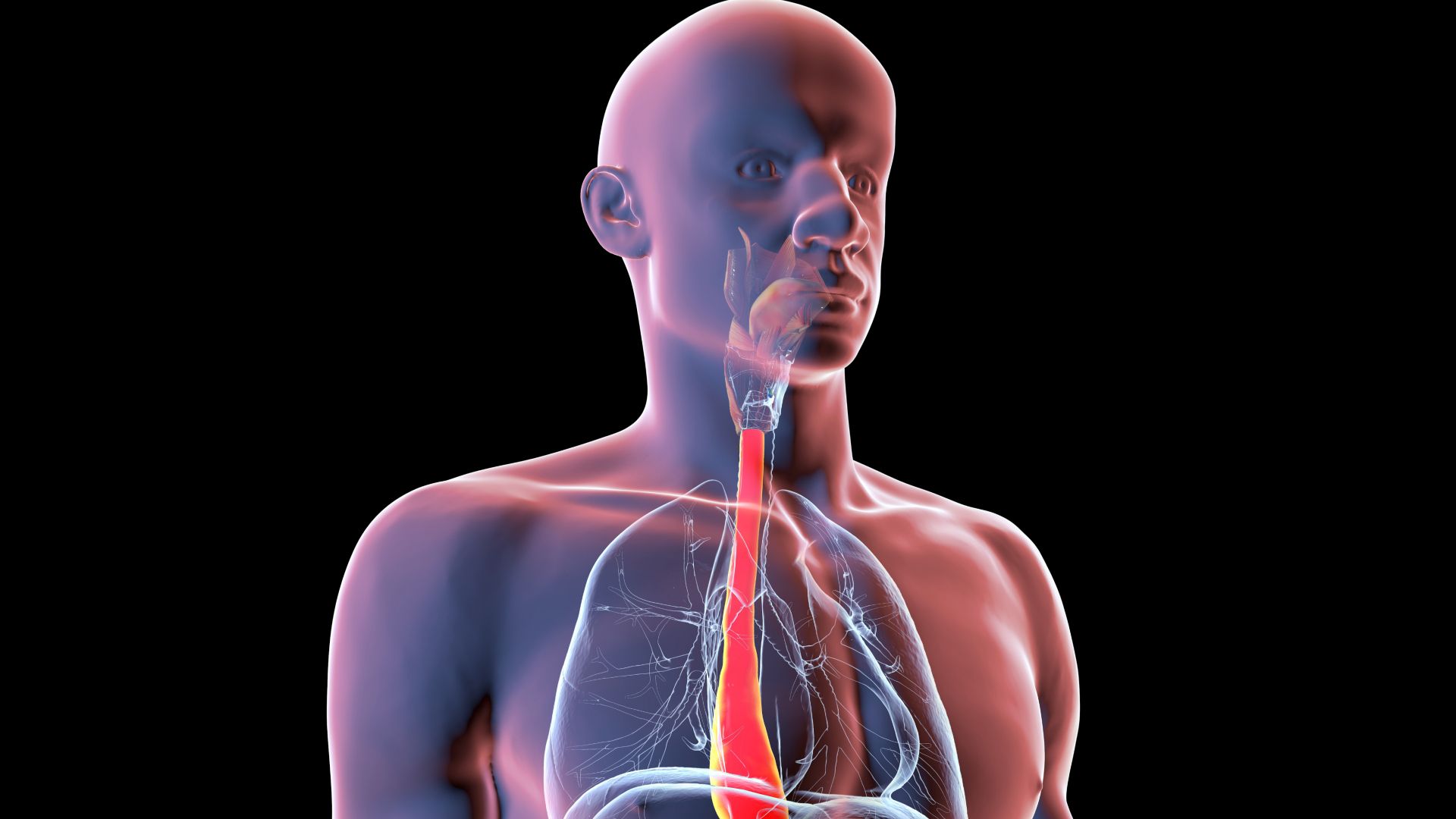
Dr. John Pandolfino jokes that he comes from a long line of electricians and plumbers — and that as a gastroenterologist, he’s stayed in the family business.
That’s because the esophagus, the organ that brings food from your mouth to your stomach, is basically a pipe with some electrical wiring, he said.
Tia Ghose: You’re using a digital twin in a condition called achalasia. Can you tell me more about what this condition is?
John Pandolfino: The esophagus, you know, its main job is to push things down into your stomach once it enters the esophagus. And then when something refluxes up, the esophagus also has to push it down to protect you from aspirating and getting it into your lungs. What happens in achalasia is, the lower esophageal sphincter, which is kind of the barrier that separates the esophagus and the stomach, doesn’t open up. It doesn’t relax. Achalasia actually means failure to relax. When that muscle doesn’t relax and open, the food just accumulates in your esophagus and you literally, almost, start drowning on your own saliva and food. So it could be a deadly disease.
What was interesting was that we noticed after treating patients, they were developing this diverticulum [a weakening and ballooning of the wall], and we didn’t really understand why this was happening. So we asked the mathematical model, the virtual esophagus — we actually gave it a bunch of options. We changed a lot of the variables, like what type of surgery are they having? How long did they cut the muscle? Did they include something called an anti-reflux procedure, where you kind of take part of the stomach and wrap it around the esophagus so you wouldn’t get reflux? Would it matter what kind of motility problem they had? There’s different subtypes of achalasia. So would some subtypes be better than others? We went through this whole process. How deep would you cut the muscle? And we just ran simulations.
So it took a couple months of training this and letting it run through millions and millions of scenarios to show what would happen. And in the end, the model actually predicted what would be the best surgery, and it also predicted which patients would be at highest risk to develop the complication.
So with that information, we then submitted an NIH grant that focused on looking at two different types of surgery: the standard approach versus one that’s modified by the virtual esophagus, so what the virtual esophagus picked. So we are going to test this standard approach, which works pretty well, versus this other approach. And we believe we’ve modeled the study so that they look equivalent, but we believe the new one will have less reflux and less diverticulum development.
TG: What you’re describing seems like a far cry from what people are describing as a canonical digital twin, where you integrate all the key chemicals and signaling processing involved, all the mechanical forces and all the real-time data from wearables and medical imaging. How far away do you think we are from that type of digital twin?
JP: From a mechanical standpoint, I think it’s pretty good already.
In terms of getting into the molecular structure and the actin [muscle filaments] and how muscles contract and the calcium influxes, I think that we’re really far away. We’ve just learned how the proteins fold; developing a mathematical model of the cell is going to take a pretty long time.
But I think, mechanically, we can do this, and the great thing is that this approach canbe adopted across all organ systems — the bladder, the aorta, the left ventricle. These processes where you rely completely on the mechanics of transport — now, we can take this and apply it across those [systems].
TG: So what you envision right now being pretty close is mainly for pump-and-tube-type systems and largely for surgeries. Do you see it as having prognostic or diagnostic value?
JP: It certainly will have prognostic value because you get to the point where you can pick up scenarios where medications will have no effect anymore, right? So, if someone gets to the point where they have deformation of the wall, that wall is gone. No medicine you give them is going to make them better.
TG: People have floated the idea that a digital twin could be used to supplant some animal research and clinical trial data.
JP: Yeah.
What it will do is, it’ll take us away from using animals for surgery.
John Pandolfino, chief of gastroenterology and hepatology and director of the Northwestern Medicine Digestive Health Institute
TG: Do you actually think that’s realistic?
JP: If you’re looking at surgeries, you wouldn’t need to do this on animals. You would do this on a simulation, like we have, to see what the effects are, and then you could actually go from that to various changes in humans. That’s exactly what happened here: Our virtual esophagus proved what we thought would probably be the right way to do this. So it proved our hypothesis mathematically, and now we’re embarking on a human trial.
TG: But most animal research, I would guess, is focused on testing new compounds that could have therapeutic potential, right? So, do you think it has much potential there?
JP: Those studies where they give a mouse like 50 times the dose that a human would get to see, is it going to kill it? I don’t think [digital twin technology will] affect that. What it will do is, it’ll take us away from using animals for surgery.
In addition, I do think that this is going to lead us to an area where we will be able to create much better models for simulation. So we’ll understand a lot more regarding the material properties of the organs, how they respond to stress and strain, and develop simulations that run not just in the virtual world but actually have tactile twins, right? So something that actually is made from a material that simulates the esophagus or simulates an organ almost perfectly so that when you’re cutting it, it’s the same feel.
TG: You could train on it and you could pour goop down it and see how it expands or something?
JP: Exactly. But you know, there’s just a lot you can learn from understanding one part of the human anatomy and function, because the body doesn’t come up with totally different ways to do things. It repeats it, and it just may make it bigger or smaller, use a slightly different length. [Organs like the bladder and heart] all work pretty similarly. They have a tube that has some contraction. There are sphincters that open up and close. If you even look at the esophagastric junction, the valve that’s at the anti-reflux barrier, it’s very similar to the ano-rectal junction where you have a bowel movement. And, in fact, if you look at the physiology of how you have a bowel movement and how you swallow and protect yourself from having reflux, they’re literally just reversed.
TG: Nature just copies itself.
JP: Yeah.
TG: So you think this has a lot more applicability across the body?
JP: Yeah, even in the esophagus. I mean reflux heartburn affects like a fifth of the country. And really, reflux is not an issue of too much acid. Most people who have reflux have normal acid.
It’s more of an anatomy and a physiology issue. So, you know, our approach will allow us to hopefully modify a lot of the surgeries that are being done for reflux and even maybe help create less-invasive approaches that work. So, just in the GI [gastrointestinal tract] in general, in the esophagus, it’s going to have a lot more application. And then even in people who may have bladder issues, an overactive bladder or maybe a hypoactive bladder, how do you assess that in relationship to flow and emptying of the bladder? [It’s] similar in aortic aneurysms. Aortic aneurysm is basically a diverticulum. It’s just a pressure-related change in the anatomy where it basically balloons out. And when it balloons out, because it balloons, it loses its function, and then the blood’s not pumping right.
This article is for informational purposes only and is not meant to offer medical advice.